Built for Compliance. Proven for Reliability
Winpak delivers a comprehensive healthcare packaging portfolio designed to meet the
diverse and highly regulated primary packaging needs of the medical and
pharmaceutical markets. From advanced flexible packaging and lidding solutions
to packaging components such as
Product Inserts (PIs), Outserts, MedGuides, Instructions
for Use (IFUs) and Pressure-Sensitive Labels, these products are essential elements of product quality,
regulatory compliance, and supply chain continuity. The components must meet
rigorous technical standards while supporting efficient operations across
complex manufacturing and distribution environments.
Winpak has been a trusted provider of these critical components for decades. Leveraging
deep expertise within its North American center of excellence in Norwood, New
Jersey and supported by a customer-centric operating model, Winpak delivers
dependable, compliant, and responsive solutions to pharmaceutical
manufacturers, biotechnology companies, and contract development and
manufacturing organizations (CDMOs) across North America.
Product Inserts and Outserts: Precision Manufacturing for Regulated Applications
Using modern, high-precision equipment, Winpak manufactures
large-format printed sheets and converts into compact inserts suitable for
automated application onto bottles, into cartons, and other pharmaceutical
packaging formats, meeting strict dimensional and FDA requirements. Winpak
combines printing quality and folding precision that ensures consistent line
performance for customers whose operations depend on stability and
repeatability.
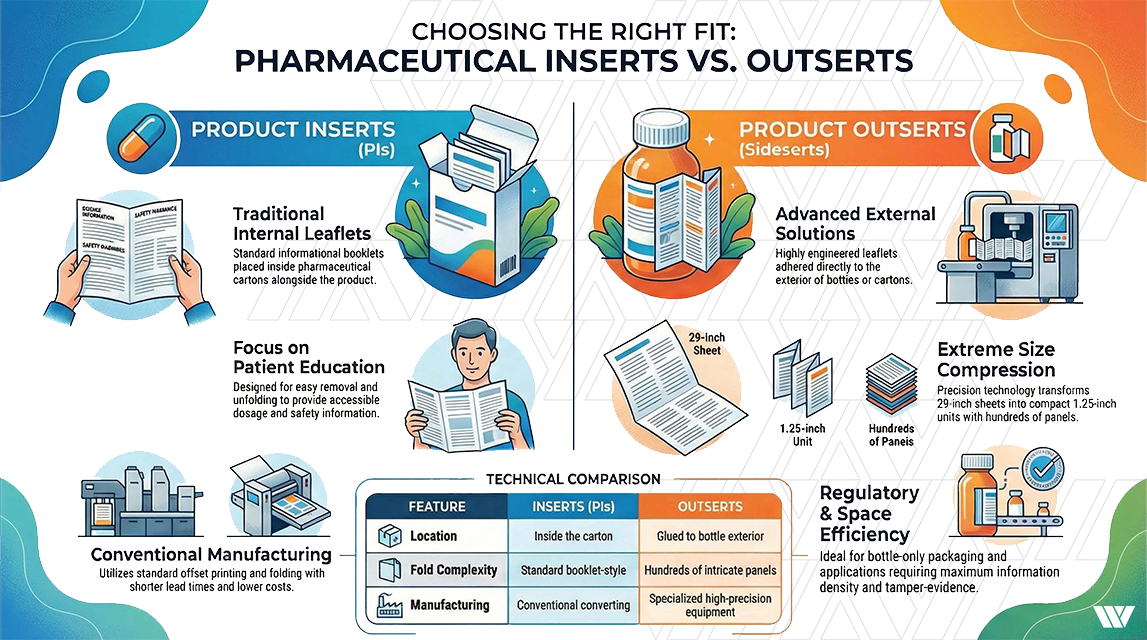
Understanding the Difference Between Inserts and Outserts
Pharmaceutical
and healthcare products rely on two key printed components to deliver essential
safety, usage, and regulatory information:
Product Inserts (PIs) and
Outserts.
While they
serve similar informational purposes, the way they are designed, manufactured,
and applied within the packaging process differs significantly.
 Medication Guides (MedGuides)
Medication Guides (MedGuides) are FDA-approved patient information
documents required for certain prescription therapies. They are designed to
communicate important safety information in a clear, accessible format, helping
patients understand serious risks, potential side effects, and proper use of
their medications.
Instructions for Use (IFUs) serve a similar role for
medical devices, combination products, and other complex therapies, providing
detailed, manufacturer-authored guidance on correct setup, operation, handling,
storage, and disposal to ensure safe and effective product use.
While MedGuides and
IFUs share similarities with Product Inserts and Outserts in purpose and
regulatory oversight, they typically involve less complex folding and format
requirements. Winpak’s experience across all four formats enables consistent
quality, regulatory
alignment, and reliable supply—allowing healthcare manufacturers to confidently
source these critical components from a single, trusted partner.
Healthcare Labels: Application-Specific Solutions with Operational Agility
Winpak’s healthcare label offering
includes pressure-sensitive labels designed for a diverse set of applications,
including plastic bottles, glass vials, pre-filled syringes, and folding
cartons. Multiple material and adhesive options are available to support ambient,
refrigerated, or specialized storage conditions, ensuring suitability across a
wide range of drug product profiles.
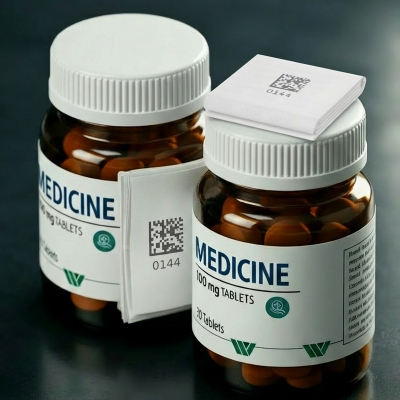
Winpak has
demonstrated flexibility in supporting customer-specific requirements such as
color matching, barcoding, serialization, and specialty constructions including
clear labels for syringe applications and labels incorporating unique ink
systems. This ability to address niche technical needs has been noted by
customers as a differentiator in the market.
Operational
responsiveness is another area where Winpak consistently performs. Customers
frequently highlight Winpak’s agility in adjusting order schedules,
accommodating compressed timelines, and supporting rush requirements, including
new product launches, when needed. This responsiveness extends to Winpak’s
ability to help organizations maintain coverage across their manufacturing
footprint, even as production schedules shift or expand.
Dependable Supply Chain Support for High-Stakes Healthcare Operations
Winpak’s focus on reliability and service has consistently translated into measurable
supply chain performance and has maintained best-in-class on-time, in-full
(OTIF) delivery levels for our healthcare customers. This level of
dependability has been particularly valued in support of critical product
launches and urgent operational scenarios, where packaging availability
directly influences production continuity.
Operational
Simplicity and Service Excellence in a Demanding Marketplace
While the
label and insert markets are highly competitive, Winpak differentiates itself
through its unmatched service model. Customers consistently recognize Winpak
for:
- Reliable supply performance that supports tight operational
timelines.
- Responsiveness and flexibility in adjusting production
schedules and managing rush requests.
- Strong distribution coverage across customer manufacturing
networks.
- Consistent communication and proactive engagement
throughout the order cycle.
- Experts in quality and
compliance expectations,
ensuring predictable and compliant outputs.
These
attributes position Winpak as a high-value partner for healthcare companies
seeking not only technical capability but also a supplier aligned with their
operational and regulatory demands.
To learn
more about Winpak’s capabilities or discuss an upcoming packaging project,
connect with our
Healthcare team.

Author
Don Alip
Product Manager, Healthcare
Sauk Village, IL
don.alip@winpak.com
Submit a comment